Denmark possesses a robust pharmaceutical industry, heavily focused on research and development and manufacturing of high-value, specialized pharmaceuticals. This creates a substantial demand for high-quality pharmaceutical intermediates and fine chemicals. The country’s stringent regulatory environment, coupled with a strong emphasis on sustainability, necessitates reliable suppliers adhering to the highest quality standards.
Geographically, Denmark’s location within Europe provides excellent logistical advantages for both import of raw materials and export of finished products. The climate is temperate, supporting consistent manufacturing operations. Economically, Denmark is a high-income nation with a skilled workforce and significant investment in the healthcare sector, driving continued growth in the pharmaceutical chemical market.
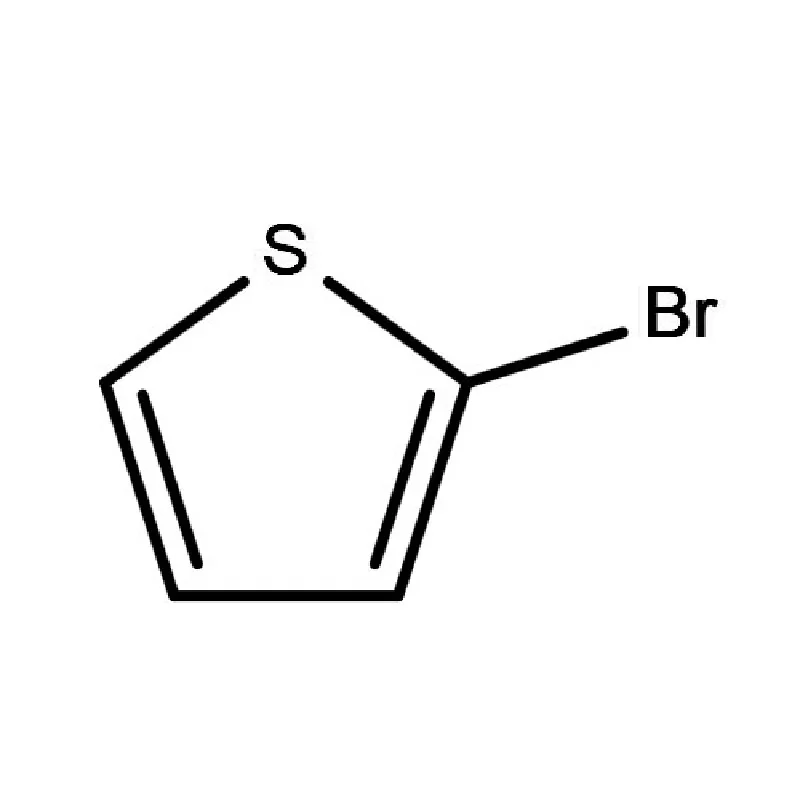
The Danish pharmaceutical sector is particularly focused on innovation and personalized medicine, leading to increased demand for complex 2 Bromothiophene derivatives and specialized chemicals used in advanced drug synthesis. This demand also extends to effective industrial water treatment chemicals ensuring regulatory compliance and environmental responsibility.