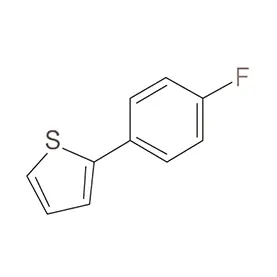
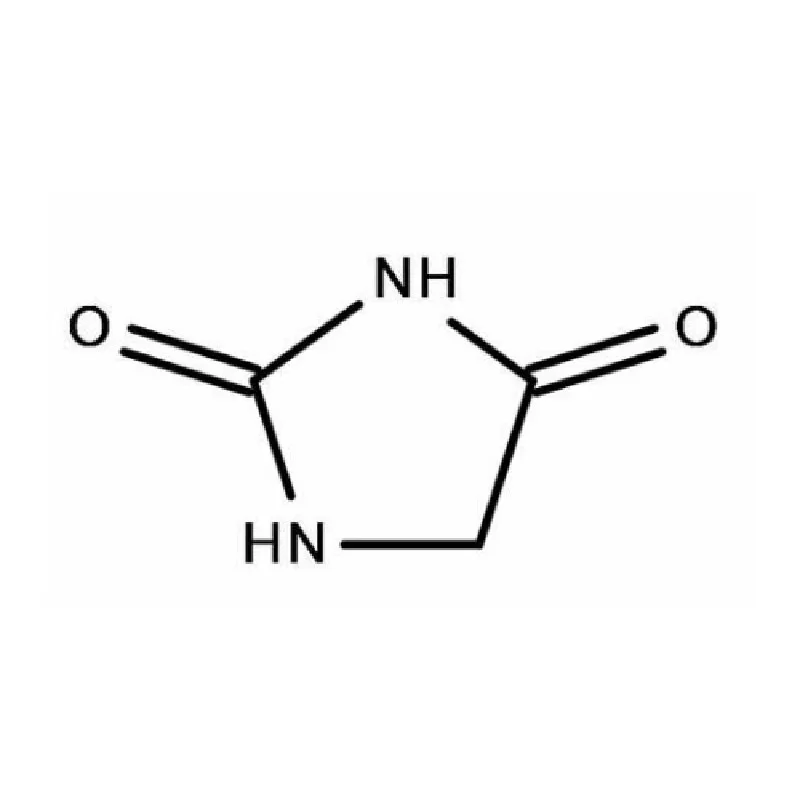
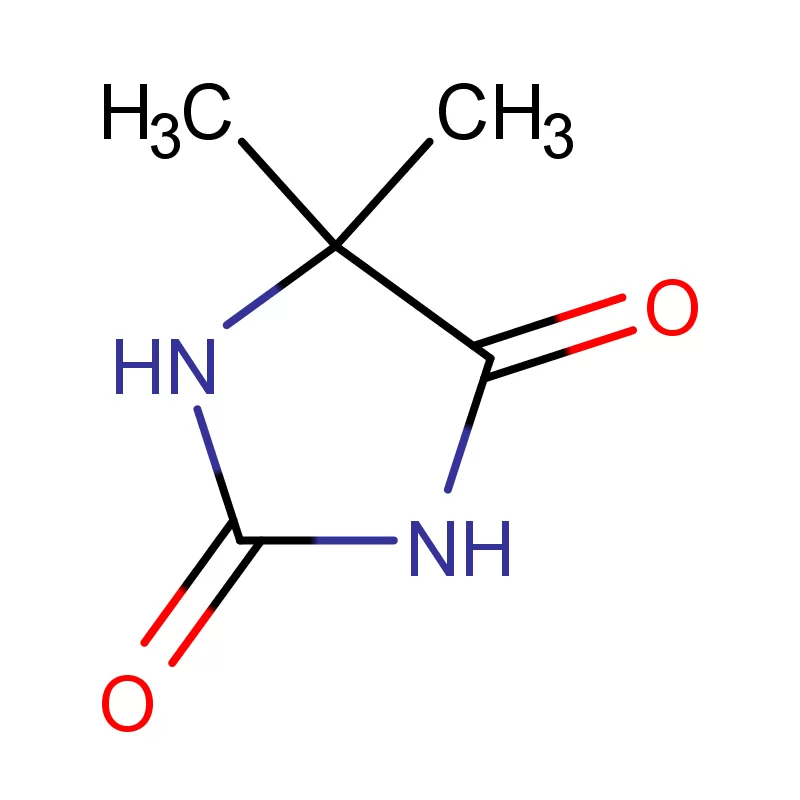
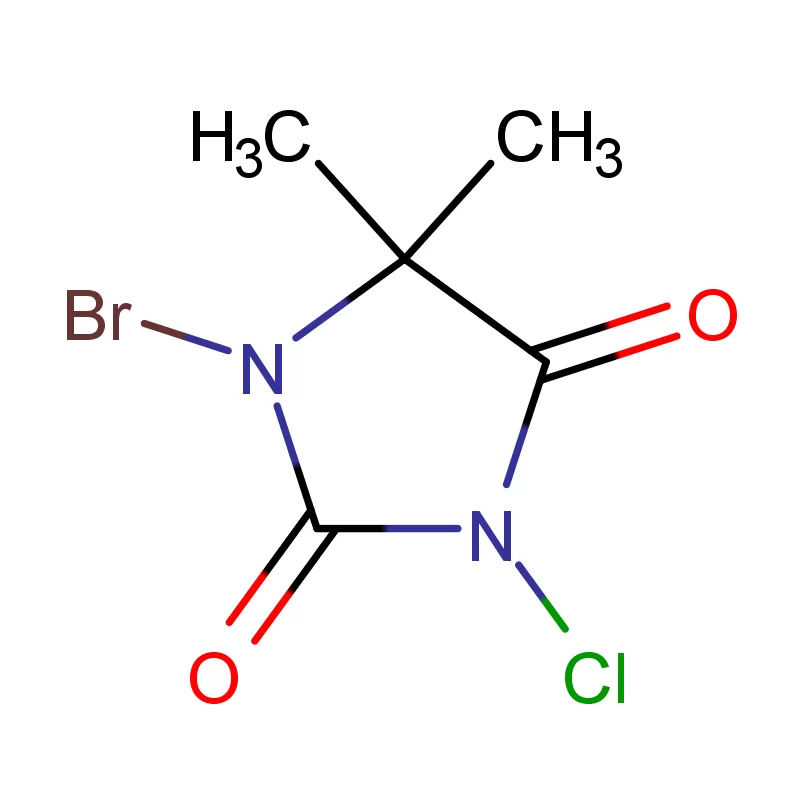
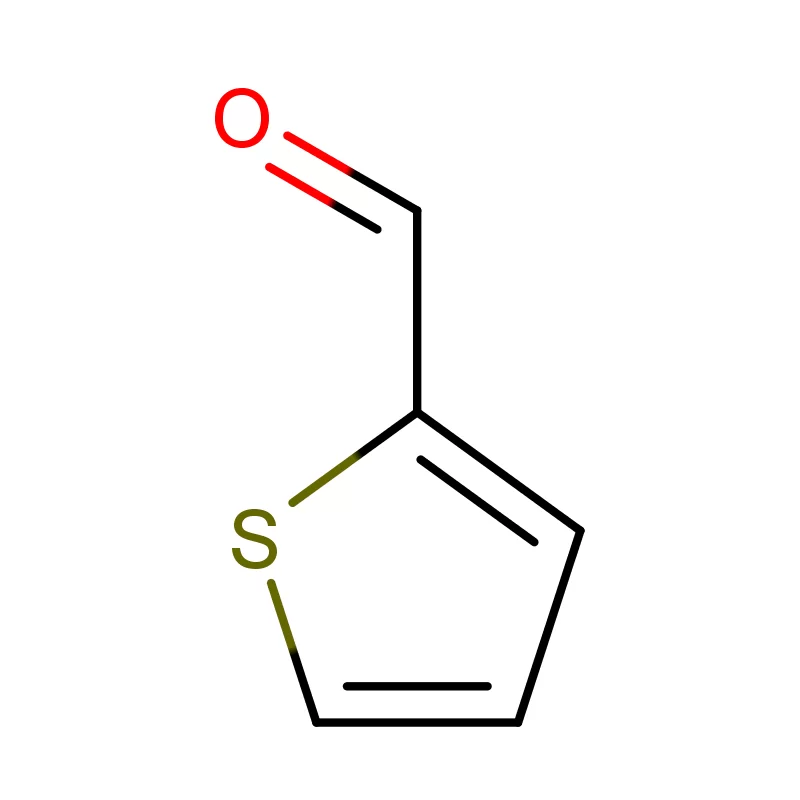
Sweden boasts a robust pharmaceutical industry, heavily reliant on innovation and high-quality chemical inputs. The country's commitment to sustainable practices and stringent regulatory standards, particularly concerning pharmaceutical intermediates, creates a specialized demand for reliable suppliers. This demand is further influenced by Sweden’s strong research and development sector.
Geographically, Sweden benefits from a strategic location within Europe, facilitating efficient logistics and distribution. The relatively cool climate requires carefully controlled storage and transportation of fine chemicals, adding another layer of complexity for suppliers. Economic stability and a skilled workforce are key strengths contributing to the sustained growth of the sector.
The Swedish market places a premium on environmentally responsible production. Consequently, companies focusing on green chemistry and sustainable sourcing of 2 Bromothiophene are particularly well-positioned to succeed. Collaboration between pharmaceutical companies and chemical suppliers is crucial for navigating the evolving regulatory landscape.