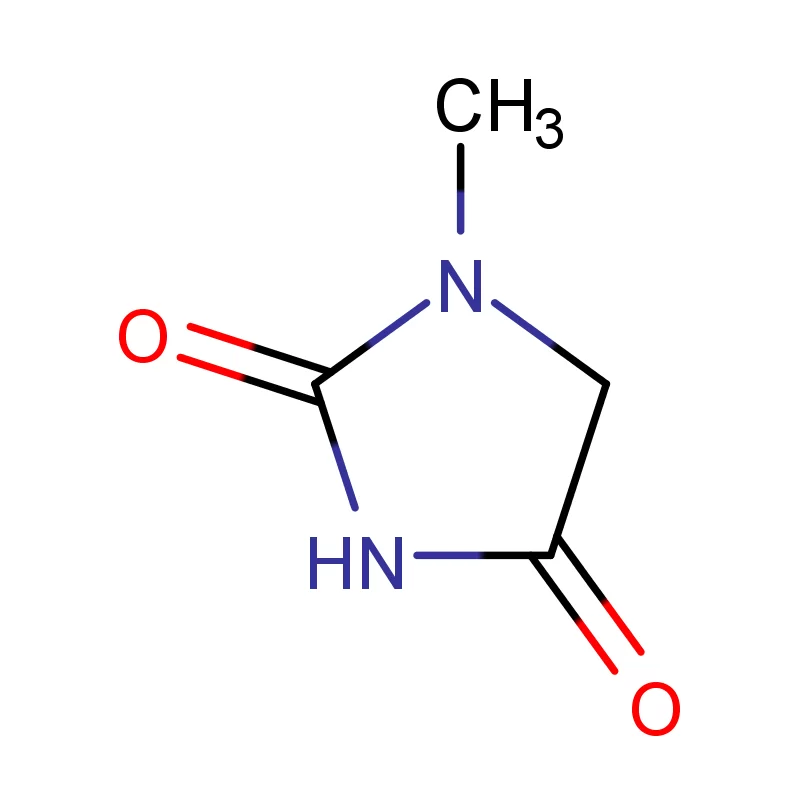
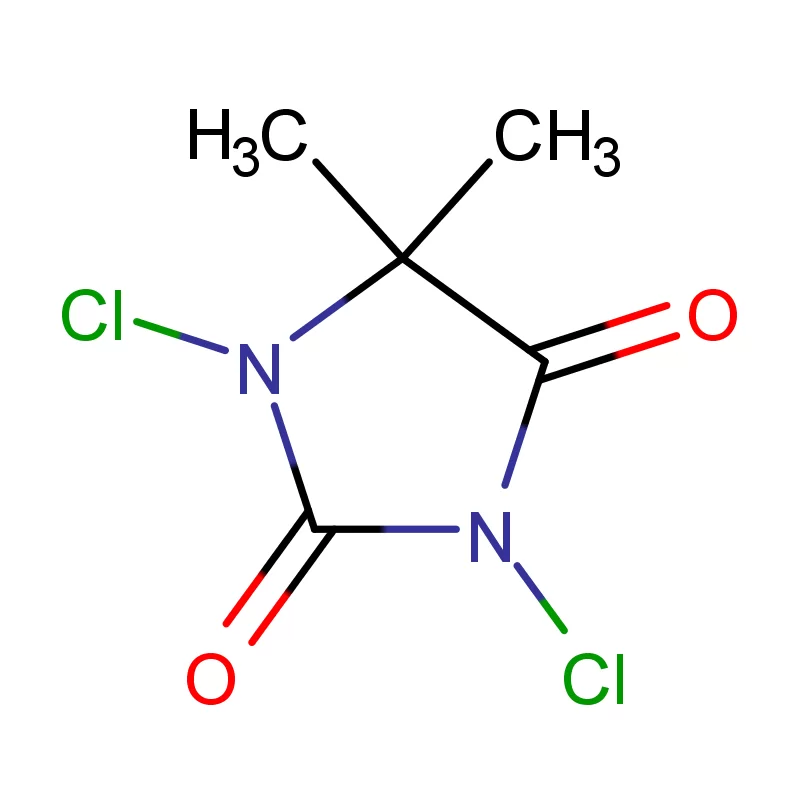
Peru's pharmaceutical industry is experiencing steady growth, driven by an expanding population, increased healthcare access, and rising disposable incomes. The demand for pharmaceutical intermediates is increasing as local manufacturers seek to enhance their production capabilities and reduce reliance on imports. However, the industry faces challenges related to regulatory hurdles and the availability of skilled labor.
The geographical diversity of Peru presents logistical challenges for the distribution of fine chemicals and raw materials. The Amazon rainforest and the Andes mountains require specialized transportation solutions, impacting costs and delivery times. Furthermore, the country's susceptibility to natural disasters necessitates robust supply chain management practices.
Peru's commitment to improving public health and expanding healthcare coverage is creating opportunities for pharmaceutical companies. The government is actively promoting investments in the pharmaceutical sector through incentives and streamlined regulatory processes. This positive environment is attracting both domestic and foreign investment, fueling industry expansion. The need for industrial water treatment chemicals is also growing with increased manufacturing.