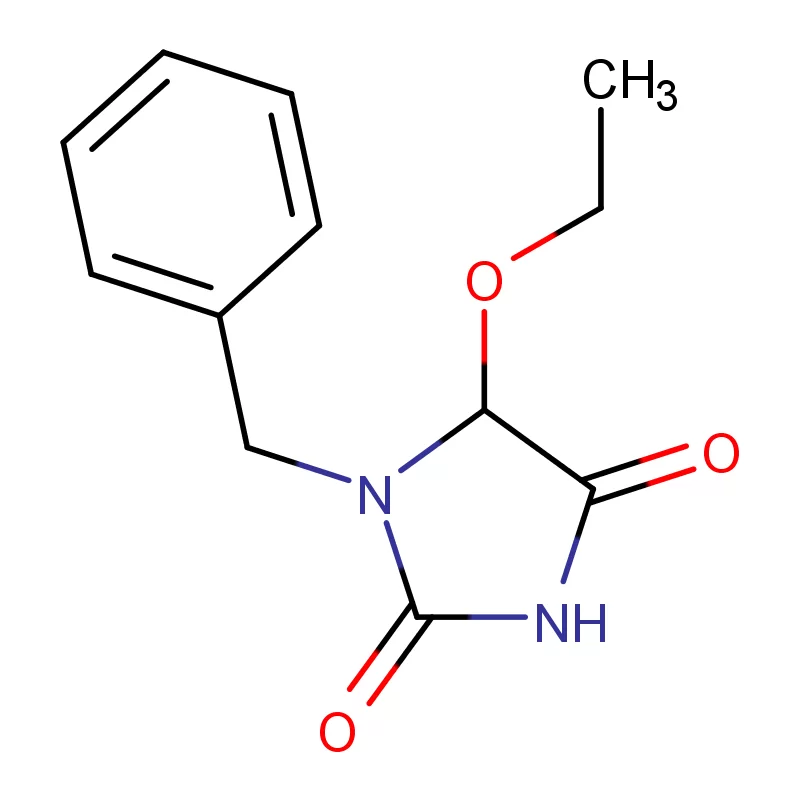
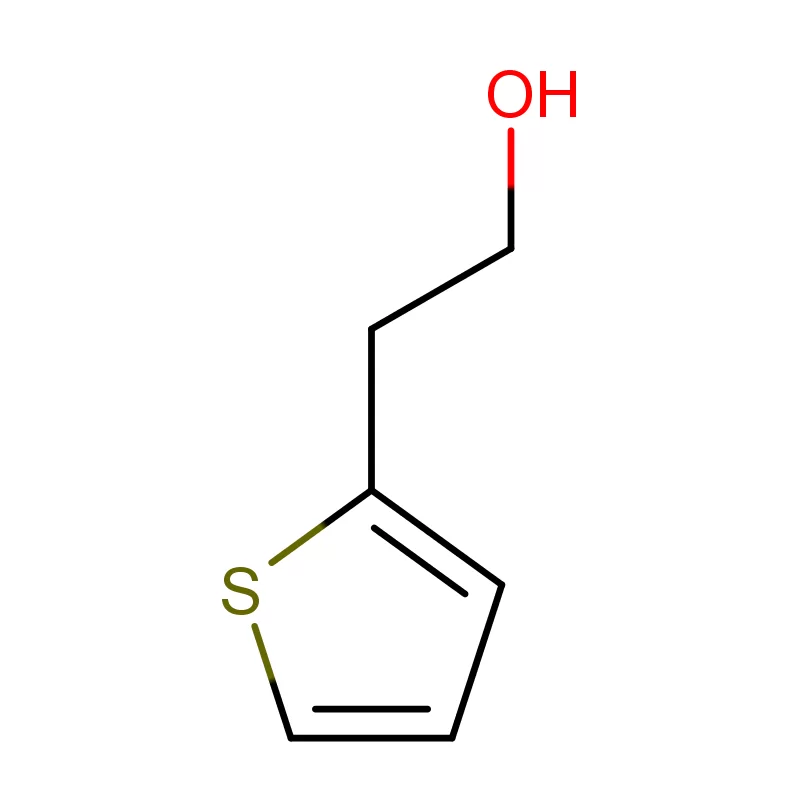
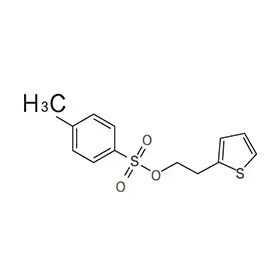
The Netherlands boasts a robust pharmaceutical sector, driven by strong research and development, a skilled workforce, and a strategic location within Europe. Demand for pharmaceutical intermediates is consistently high, fueled by both domestic manufacturing and the export of finished products. The country's commitment to sustainability also creates a demand for greener chemical processes.
Geographically, the Netherlands' access to major ports like Rotterdam facilitates efficient import and export of fine chemicals. Climate control is less of a concern compared to other regions, allowing for consistent production and storage. Economically, the Netherlands offers a stable and attractive environment for chemical companies, with government support for innovation.
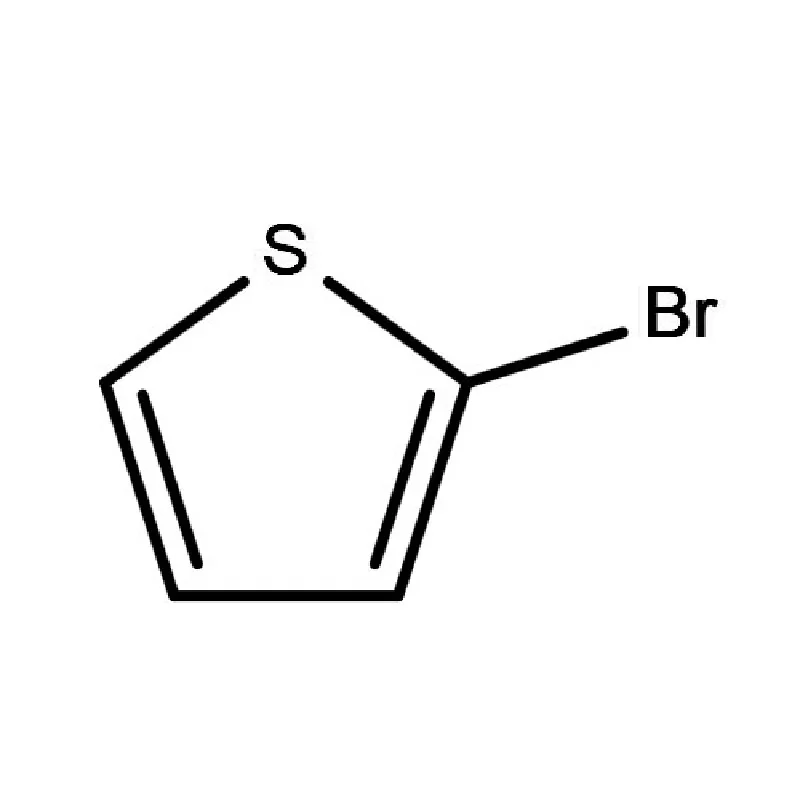
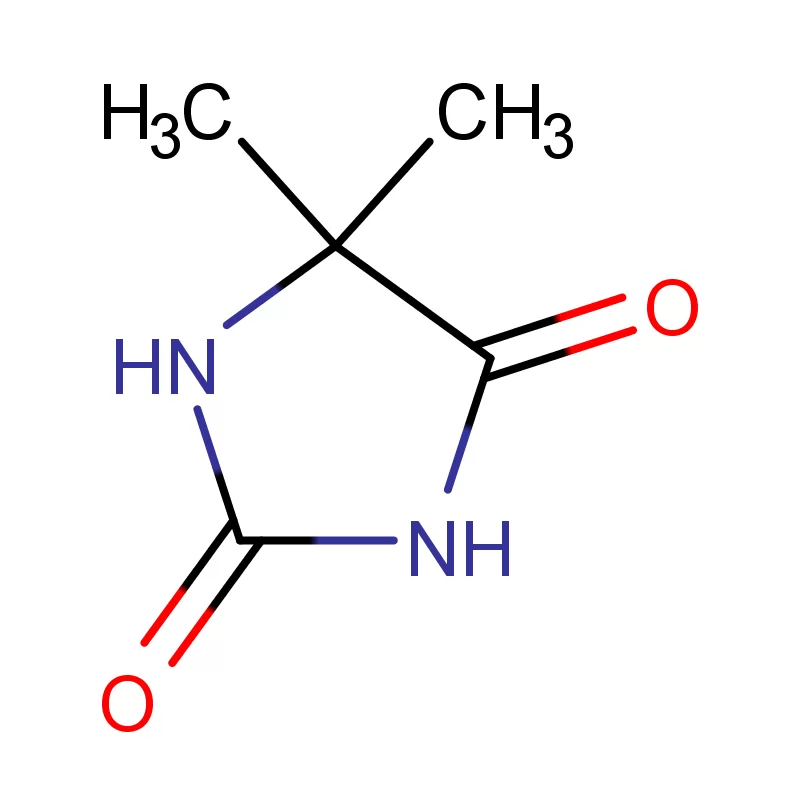
The Dutch pharmaceutical industry heavily relies on dependable supply chains for critical raw materials. Companies prioritize quality, compliance, and cost-effectiveness when sourcing 2 Bromothiophene and other key ingredients. Increasingly, they are looking for suppliers who can offer custom synthesis and specialized chemical solutions.