Switzerland boasts a highly developed pharmaceutical sector, renowned for its innovation and stringent quality control. The country's geographic location, coupled with a strong emphasis on research and development, makes it a key European hub for pharmaceutical production and industrial water treatment chemicals. The competitive landscape demands reliable sourcing of high-purity pharmaceutical intermediates.
The Swiss pharmaceutical industry is heavily regulated, adhering to both Swiss national standards and European Union guidelines. This translates to a high demand for suppliers who can demonstrate consistent quality, traceability, and compliance. Climate stability allows for consistent production and logistical efficiencies, however, costs associated with operating in Switzerland are notably higher than in many other European nations.
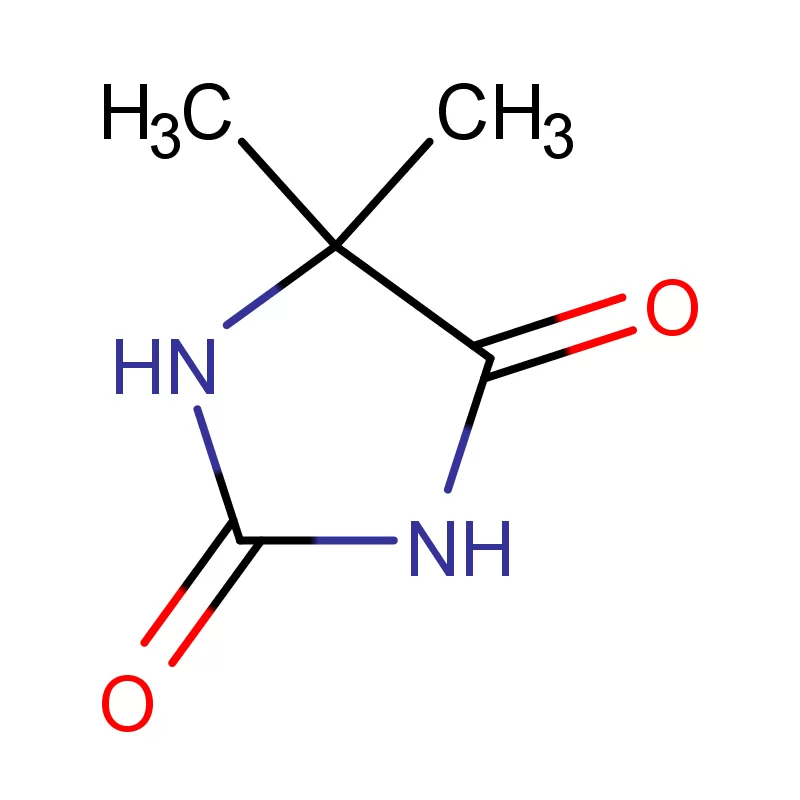
The local focus on generics and biosimilars is growing, increasing the need for cost-effective, yet high-quality, raw materials. Environmental sustainability is also a significant factor, with increasing pressure on manufacturers to adopt green chemistry practices and responsible waste management. The demand for 5 5 Dimethyl Hydantoin and other specialized chemicals remains robust.