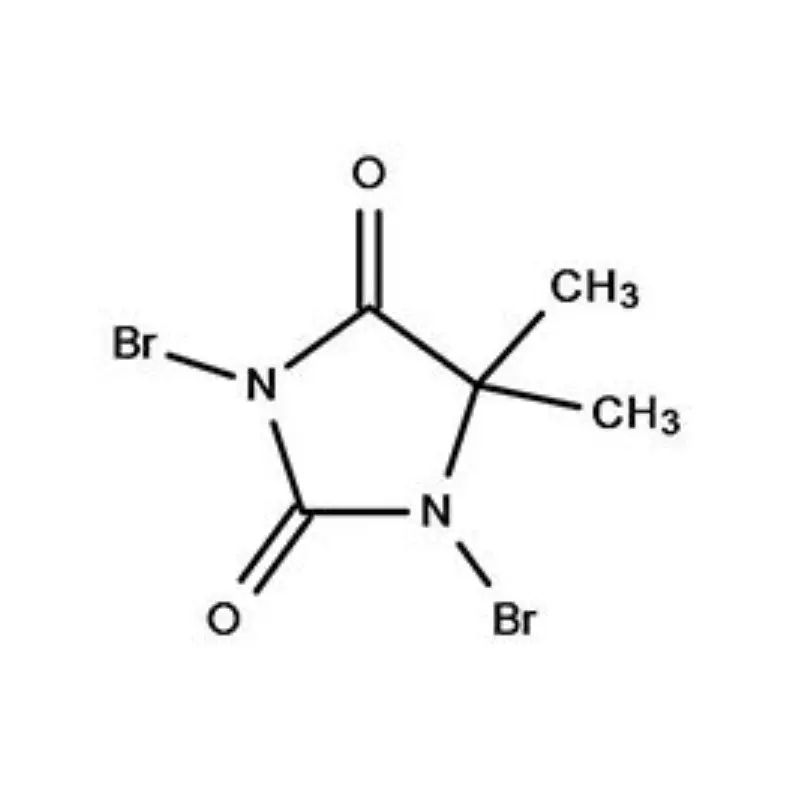
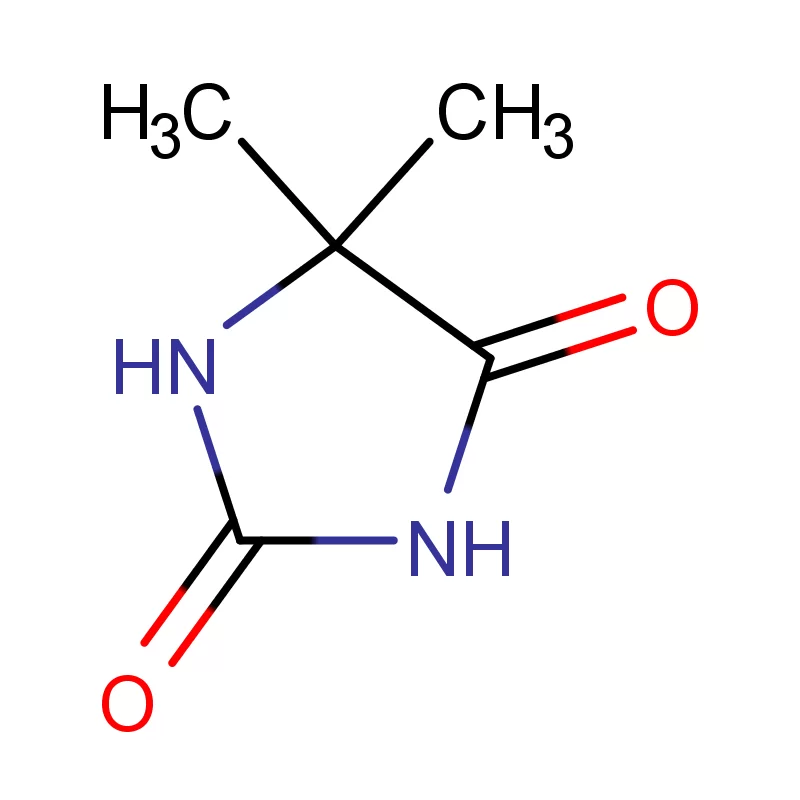
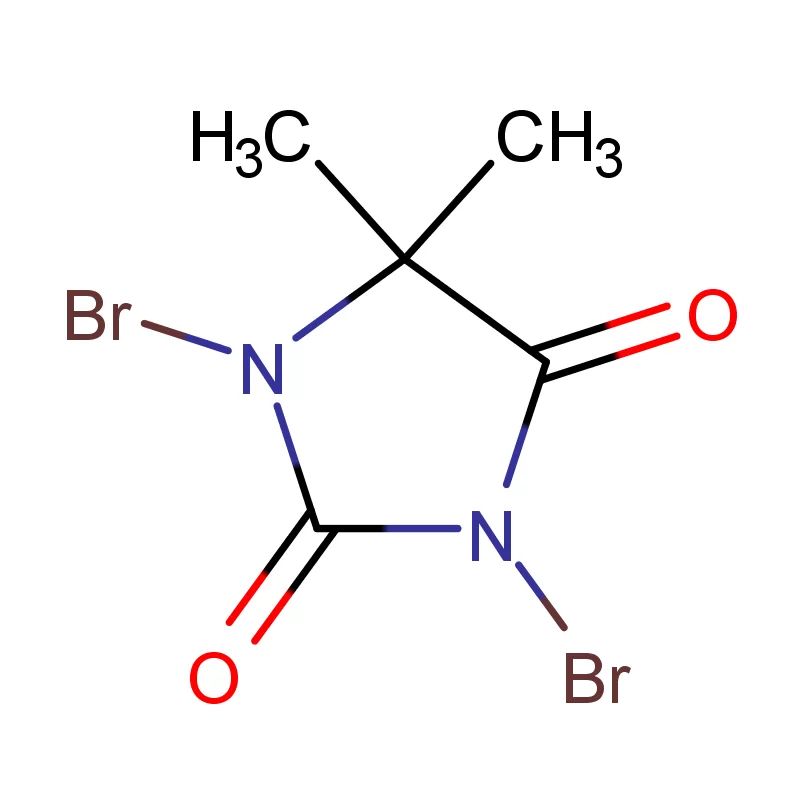
Cambodia's pharmaceutical industry is currently experiencing significant growth, driven by increasing healthcare awareness and government initiatives to improve access to essential medicines. The reliance on imported pharmaceutical intermediates is high, presenting both opportunities and challenges for local manufacturers. The tropical climate necessitates careful storage and transportation of sensitive chemicals, adding complexity to the supply chain.
The regulatory framework for pharmaceuticals in Cambodia is evolving, with increasing emphasis on quality control and adherence to international standards. Demand for fine chemicals is rising as local companies seek to enhance their manufacturing capabilities and reduce dependence on imports. Access to financing and skilled labor remains a key constraint for many pharmaceutical businesses.
Water quality is a critical factor in pharmaceutical manufacturing, and the need for effective industrial water treatment chemicals is growing to ensure compliance with stringent quality requirements. The increasing focus on generic drug production further fuels the demand for cost-effective and high-quality raw materials.