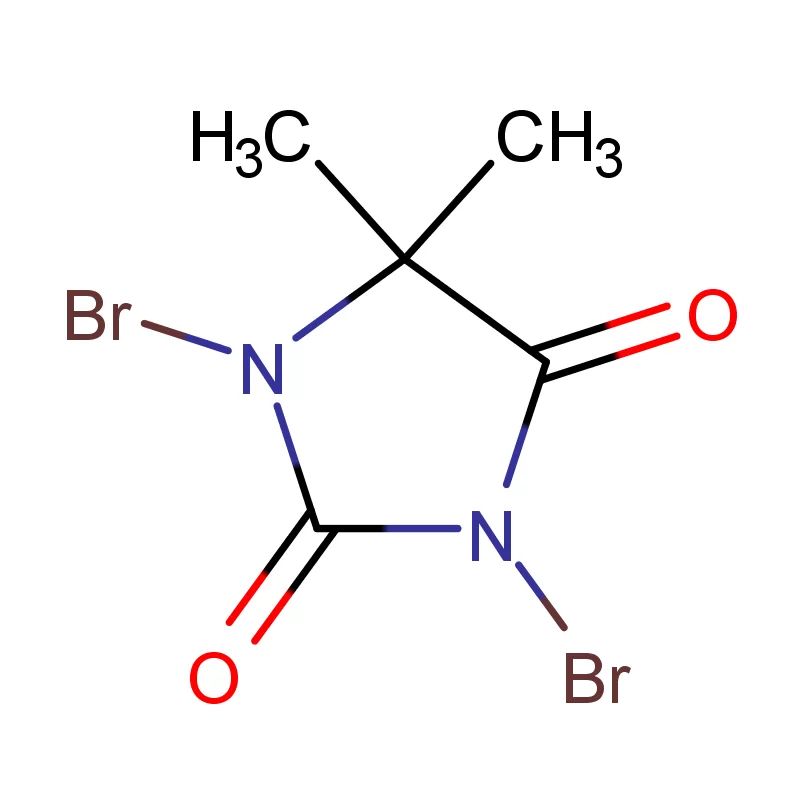
Portugal's pharmaceutical industry is a growing sector, driven by increasing healthcare demands and a focus on generic drug manufacturing. This growth necessitates a reliable supply of high-quality pharmaceutical intermediates. The geographical location of Portugal, as a gateway to European and African markets, plays a pivotal role in its pharmaceutical trade.
The climate in Portugal allows for efficient transportation and storage of chemical compounds, which is crucial for maintaining product integrity. However, stringent EU regulations regarding chemical manufacturing and environmental protection present both challenges and opportunities for companies operating within this sector. Fine chemicals demand is specifically impacted by these regulations.
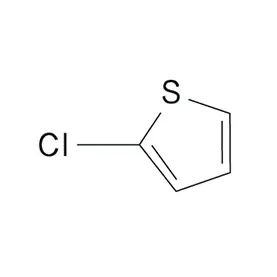
Portugal's economic stability, compared to some other European nations, offers a relatively secure investment environment for chemical manufacturers. The country's skilled workforce and access to EU funding further contribute to the growth of the pharmaceutical intermediates industry. Demand for 2 Bromothiophene is steadily increasing.